AstraZeneca’s COVID-19 vaccine begins phase 3
AstraZeneca, the biopharmaceutical company, said its COVID-19 vaccine AZD1222 expands into a phase 3 clinical trial in the U.S., enrolling up to 30,000 adults. Meanwhile, the company reported that its Imfinzi has been approved in the European Union for the treatment of extensive-stage small cell lung cancer. Also, it said its Farxiga demonstrated unprecedented reduction in the risk of kidney failure and cardiovascular or renal death in patients with chronic kidney disease in the Phase 3 DAPA-CKD trial.
From a chartist’s point of view, the stock price has entered into a consolidation move and is capped by the key resistance threshold at 8811p. The 50-DMA started to flatten out indicating a lack of momentum in the short term. The Daily Relative Strength Index (RSI, 14) is capped by a declining trend line. Readers may want to consider opening Short positions below the key resistance level at 8811 with 8048p and 7685p as targets (congestion zone). Alternatively, a push above 8811p would open a path to see 9400p.
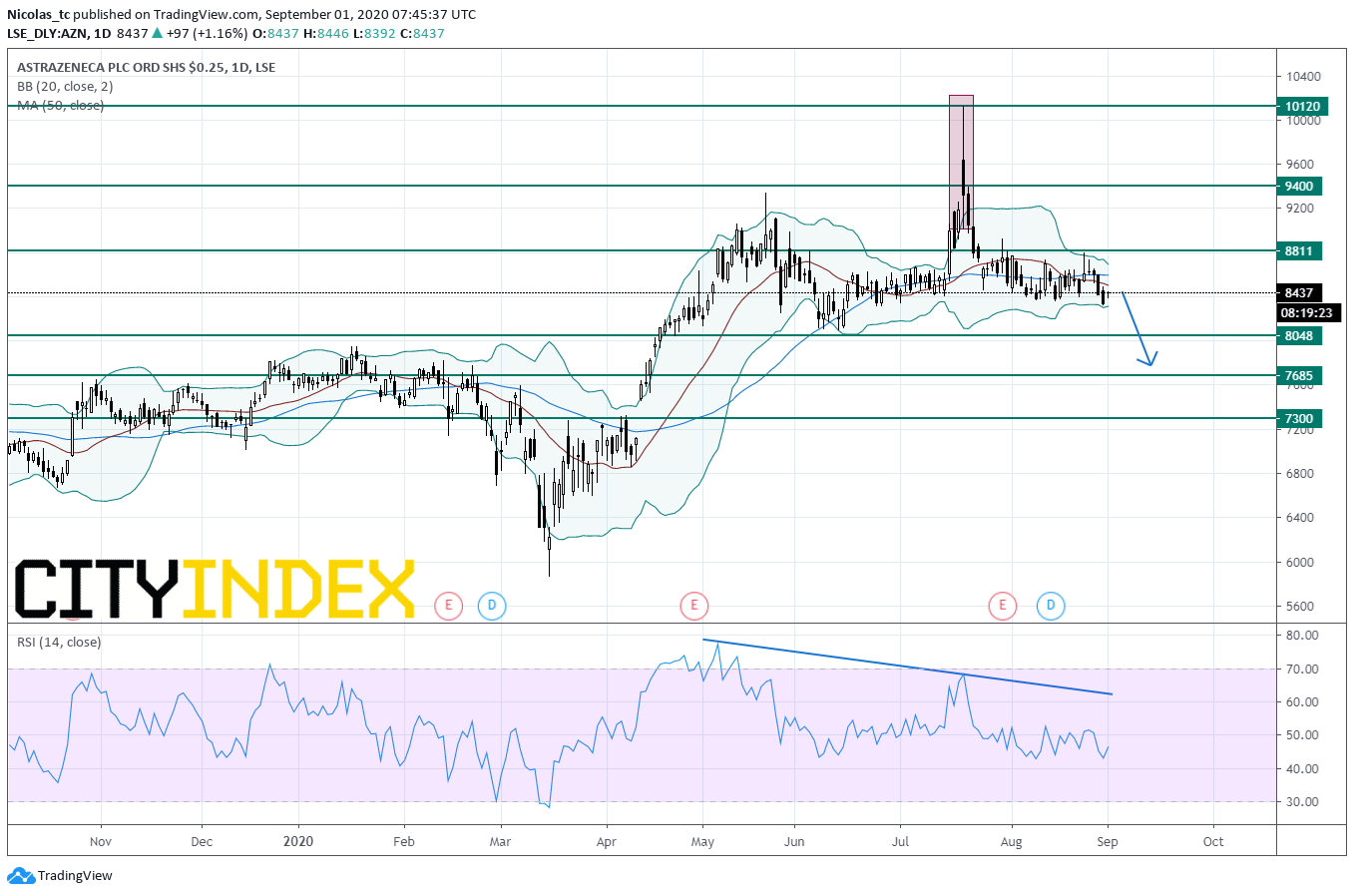
Source: GAIN Capital, TradingView
Latest market news
Today 11:14 AM
Today 08:28 AM
Yesterday 03:30 PM
Yesterday 01:23 PM
Yesterday 11:00 AM