AstraZeneca : positive review from FDA
AstraZeneca, a global biopharmaceutical company, announced that its Farxiga (dapagliflozin) has been granted Breakthrough Therapy Designation (BTD) in the U.S. for patients with chronic kidney disease.
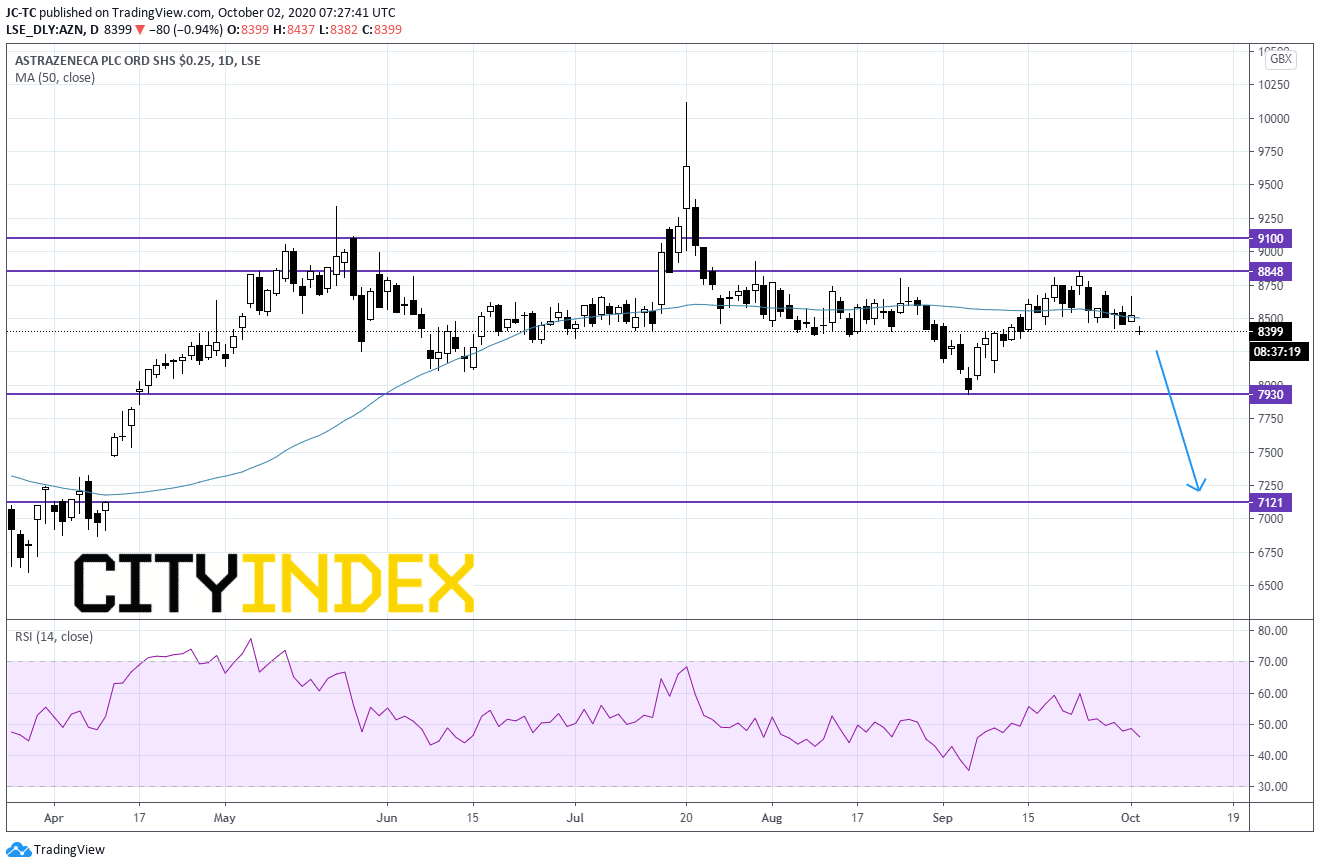
From a chartist point of view, on a daily chart, prices are stuck below the resistance at 8848. The 50 day MA is negatively oriented above prices while the RSI is in bearish territory. As long as the 8848 resistance is not broken, readers may therefore consider further downside towards 7930 and 7121 in extension.
From a chartist point of view, on a daily chart, prices are stuck below the resistance at 8848. The 50 day MA is negatively oriented above prices while the RSI is in bearish territory. As long as the 8848 resistance is not broken, readers may therefore consider further downside towards 7930 and 7121 in extension.
Source : TradingVIEW, Gain Capital
Latest market news
Yesterday 01:23 PM
Yesterday 06:01 AM
April 18, 2024 11:27 PM
April 18, 2024 04:46 PM

